 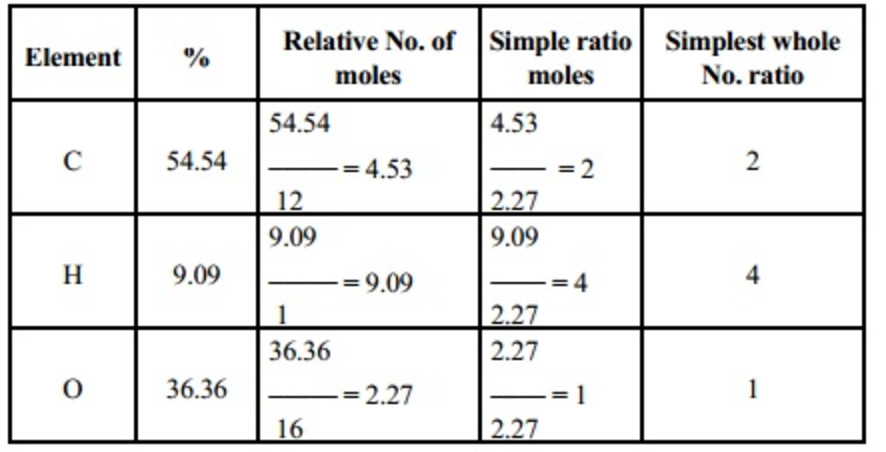
After rounding this result to a whole number we have n = 6, and obtain the molecular formula of glucose: C 6H 12O 6. Next, we divide the molecular weight of the substance under study by the just found molecular weight of the empirical formula and obtain: 180.16 / 30.026 = 5.954. Using our Molecular Weight Calculator we can easily get 30.026 g/mol. To this end, we first find the molecular weight of the empirical formula, that is, the weight in grams of one mole of a substance having the chemical formula CH 2O. Now, if we know that the molar mass of the compound under study is 180.16 g/mol, we can find the molecular formula of this compound. If we now divide the found numbers of moles by the minimum value of 0.1665 and round to whole values, then we get the following indices for the empirical formula: 1, 2, 1. integer multiples of the subscripts of the empirical formula).Ġ.336 g H / 1.008 (g/mol) H = 0.3333 mol H The molecular formula is always some integer multiple n of the empirical formula (i.e. Then we consider the number of moles of each element relative to each other, converting these numbers into a whole-number ratio that can be used to derive the empirical formula of the substance. The easiest way to express the number of atoms is in moles.įor this, we have to use molar masses to convert the mass of each element to a number of moles. So, any experimental data involving masses must be used to derive the corresponding numbers of atoms in the compound. The most common approach to determining a compound’s chemical composition is to measure first the masses of its constituent elements. To find the empirical formula of a substance you can use our Empirical Formula Calculator. The empirical formula of a chemical compound is the simplest whole number ratio of atoms present in the compound. The molecular formula should be distinguished from the empirical formula. (NH 4) 2SO 4, The first part of the formula is NH 4, the second part of the formula is SO 4.The molecular formula describes the exact number and type of atoms in a single molecule of a compound. The chemical name of the compound is calcium nitrate The chemical symbols of the Periodic Table is also included in the calculator.Ī separate 'chemical names' calculator has also been included to help students name particular chemicals or elements from their chemical formulas or symbols.Ĭa(NO 3) 2, The first part of the formula is Ca, the second part of the formula is NO 3. The sharing of two electrons produces a covalent bond. The non metals obtain a stable outer or valence electron configuration by sharing electrons with one another. Covalent compounds are composed of non-metallic elements. The chemical formula calculator also contains the names of a range of covalent compounds which occur as acids. This way students can see that the ions combine in whole number ratios in order to produce a neutral chemical species.ī) the chemical formula of the compound appears after the arrow. Potassium carbonate, 2K + + CO 3 2 - → K 2CO 3 Sodium sulfate, 2Na + + SO 4 2 - → Na 2SO 4 The electrostatic attraction between oppositely charge ions produces an ionic bond.Ī) the net ionic equation of an ionic compound Non-metals gain electrons to produce negative ions called anions. 
Metals lose electrons to produce positve ions called cations. 
The transfer of electrons between metals and non-metals produces charged particles called ions. Sodium chloride, NaCl and magnesium oxide, MgO The chemical formula of ionic compounds can be quickly calculated using the chemical formula calculator.Īn ionic compound is composed of a metal and a non-metal. The calculator can be used to calculate the chemical formula of a range of The calculator is found on the right hand panel of the main page. The chemical formula calculator - Find the formula for ionic compounds with the net ionic equations common acids and the symbols of the elements of the Periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
